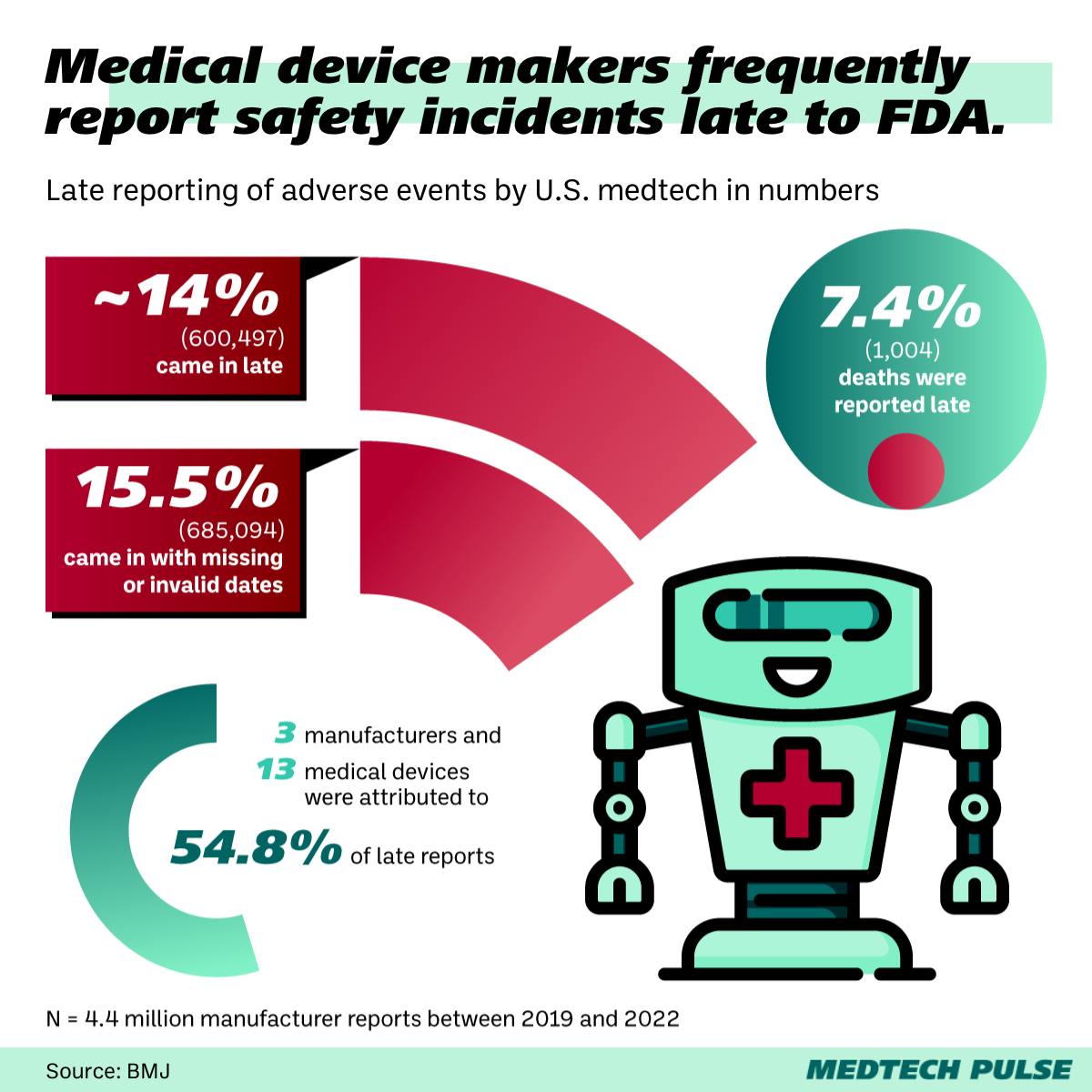
Medtech manufacturers sometimes report safety incidents late.
A new study in the BMJ highlighted how often medtech manufacturers report adverse events to the U.S. FDA later than mandated. While a majority of reports in the study period came in complete and on time, many were late and/or incomplete—including late reports of 1,004 deaths. Interestingly, just three manufacturers and 13 medical devices were responsible for over half of these late reports.