The FDA’s newest approved device: A baby sock
This isn’t your typical medical device. Owlet has finally received FDA clearance for its prescription-only smart baby sock, BabySat.
This, of course, is no regular baby sock. It’s a pulse oximeter—it tracks a baby’s pulse rate and oxygen saturation and sends alerts if the readings fall outside the target range.
The clearance comes two years after the startup received a warning letter from the FDA, for allegedly selling a medical device without having received FDA marketing clearance or approval.
Let’s take a look at how this fascinating (and very cute) device works—and what this approval means for the company.
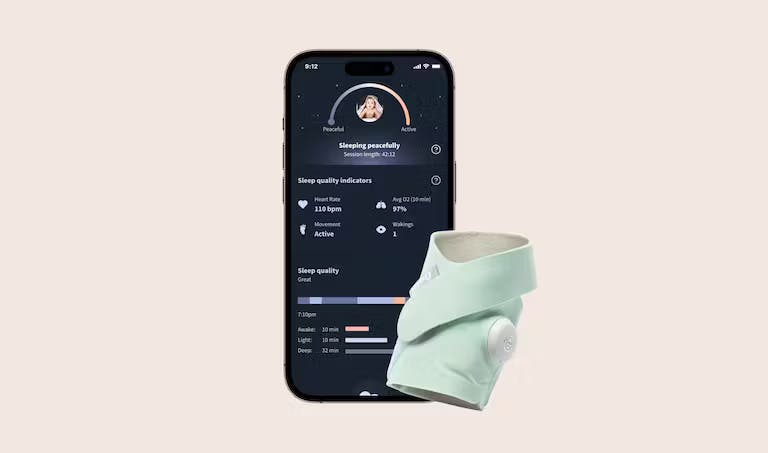
How does the smart baby sock device work?
As you can probably surmise, you won’t be buying this sock as a gift for your newborn niece or nephew.
The 510(k)-cleared BabySat device is designed for physician-supervised monitoring. Prescriptions are for infant patients who physicians believe could use additional vitals monitoring at home. The wireless sock captures pulse and oxygen saturation readings and uploads the data to an associated mobile application.
Owlet estimates that the first four years of children’s lives result in roughly 92 million infant care visits. Remote monitoring via the smart baby sock seeks to relieve the resulting strain on the availability of hospital beds and quality of care.
“Our mission is to provide caregivers with the right information at the right time to make informed decisions about their baby’s health,” said Owlet CEO Kurt Workman. “Today, parents whose babies need additional monitoring are sent home with traditional solutions that can be restrictive and more cumbersome for parents. BabySat pushes forward the modernization of hospital-grade technology for at-home use, and underscores our commitment to transforming baby care solutions.”
This device is an updated version of Owlet’s earlier device, Smart Sock, which is now only available in markets outside North America.
But is this approval enough to save Owlet?
As Owlet celebrates this key regulatory approval, it is not out of the financial woods.
The company has been dealing with the fallout from a class action lawsuit filed by one of its investors, who alleges that Owlet failed to disclose that it would need to get FDA approval for the Smart Sock device.
In the meantime, as Owlet has been unable to sell Smart Sock in the key U.S. and Canadian markets, their reported revenues have dropped 50% in the first quarter of 2023. When it launches later this year, BabySat will be available in the U.S. only.
As Owlet’s future remains to be determined, their case remains a lesson to medical device innovators. Market fit, effective product design, and even a path to profitability aren’t enough—regulatory approvals are a requisite hurdle for a large portion of our industry. As wearable health & wellness devices and AI-enabled tools may soon come to require greater regulatory oversight, more young companies in our industry are likely to face similar challenges.